
The Icelandic Arctic charr breeding programme was initiated in 1989 based on individuals from 9 different Icelandic populations (see below) and is running since. The first selected cohort was generated in 1993 and we are currently housing fish selected for up to 9 generations. The breeding station is situated in Hólar, Iceland.
In the past, fish have been selected for increased growth rate using both between- and within-family selection and for late sexual maturation using between-family selection. Recently, late sexual maturation is also selected for using within family selection. Selection proceeds via a breeding value-based index for several traits.

More recently, fish are additionally selected against high early deformity rates (expressed at first feeding and after the first few months), against high late deformity rates (expressed at the age of 2 years), and against low survival rates until the age of 2 years.


We are currently investigating whether we can increase egg quality traits, such as egg size and egg survival rate. These traits may be influnced by either, or a mix of, environmental effects (growing conditions and practices), direct genetic effects (genetic effects inherited from both parents that affect egg quality traits), or by maternal genetic effects (direct genetic effects of both grandparents on maternal traits whose expression in the mother affect quality traits of her eggs or offspring).
We found that female spawning time (ovulation date) has a high heritability in this population and we are working on improving the breeding programme via optimising spawning date variation. Spawning date variation spans many weeks and because individual females have a small time window when their eggs can be successfully fertilised, this has been limiting possible matings, and sometimes also compromised egg survival via mismatching spawning date and ovulation date, and thereby limited the overall efficiency of the breeding programme.
We also found that egg size and fecundity both have a high heritability and both are genetically correlated with body size at age 2 years (before initiating sexual maturation), and with the body size when fish mature at age 3 or 4 years. However, both maternal traits are also influenced by environmental effects and good growth conditions lead to smaller eggs but lower fecundity. Thus, selection for larger body size in combination with sub-optimal growth conditions may result in larger eggs but fewer of them, whereas good growth conditions result in smaller eggs but more of them. We are working on a publication about this topic.
Recent research on how to implement a feasible quantification method that allows selecting for increased resistance to bacteria that causes furunculosis turned out to not work under the chosen settings. Unfortunately, background bacterial load in healthy fish, which we hoped to use instead of the common practice of advertently infecting fish and monitoring temporal survival rates, was too low to allow for a meaningful analysis of load variation. However, we have not given up the hope to improve disease resistance without advertently infecting fish with a deadly diseases.
We have now first results about why body deformity rates are elevated in only some cohorts and under only some environmental conditions. Preliminary results indicate that early expressed body deformities (at first feeding and a few months thereafter) are determined by maternal genetic effects and that later expressed body deformities (at the age of 2 years) are determined by direct genetic effects. However, we do not hold adequate data to test for maternal genetic effects in the 2-year old fish. Similar breeding value rankings among families based on either maternal genetic and direct genetic effects indicate that the late-expressed deformities may also be of maternal genetic origin. It appears that having included only few breeders with extremely high maternal breeding values for early deformity rates in specific cohorts of the breeding programme has caused peaks in deformity rates 1-2 generations later. Fortunately, genetic variation for deformity rate appears not to be related to genetic variation for growth rate, so that select against high deformity rates at either age is unlikely to compromise selection for higher growth rate. We still do not understand why the later expressed body deformities vary among growing conditions in some cohorts. To understand variation among growing conditions, we sent out a short online questionaire to 10 Icelandic producers utilising our eggs. However, the participation was, unfortunately, quite low and did not yield any useful insight into environmental variation.
The Beginning
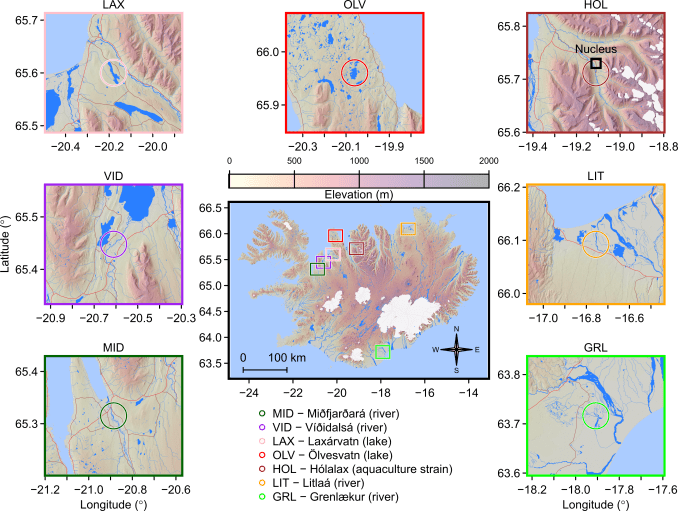
During the breeding programme foundation period (1989-2003) 13 Icelandic Arctic charr populations were evaluated via several projects and individuals from 9 populations were chosen to become broodstock. The 7 populations contributing > 3% of the genetic background to the current 9th generation are indicated in the map below. Two additional populations that are not shown contribute each < 1%. The largest contributor (47%) originates from a lake in northern Icleand (OLV) and the second largest (29%) from a small river (GRL) in southern Iceland. The location of the breeding station (Nucleus) is close the the origin of the contributing Hólalax (HOL) strain and also indicated in the map below. Beause we hold a pedigree that reaches back to the founder period, we are able to select with statistical models that account for these genetic group effects.
This page may receive updates from time to time.
